Beginners in the apiary – April
It’s never too early to think about swarming. By April, there should be lots of new bees coming through. The strongest colonies will be heaving with bees and the swarming instinct will begin to kick in once the colony starts to become crowded.
April may seem early to start swarm checks, but depending on the winter conditions and the strength of the colony, swarm preparations may already be under way. Swarm checks are usually performed every ten days or so. It is important for every beekeeper to recognise the difference between play cups, swarm cells, supersedure and emergency queen cells, and to act accordingly.

Play cups or queen cell cups, often positioned on the bottom of the frames are fairly short cup-like protrusions from the wax. They won’t necessarily develop into queen cells, so don’t worry about them.

Swarm cells will be positioned on the bottom or around the sides of the frames. They are very long and usually have some white mottling and there will be a lot of bee activity at the site. If you look inside, it will usually contain either an egg with some liquid (food) or a developing larva. During swarm checks, shake the bees off the frame and carefully survey, around the edges especially, for swarm cells. There may be ten or more swarm cells in a colony gearing up to swarm.

Emergency cells are usually found embedded more centrally on the frames but may be anywhere on the frame. Swarm cells are made with time and intention, while emergency queen cells are selected in an emergency (because the queen is dead or missing) from the eggs that are available and deemed to be appropriate to rear a queen from. Like swarm cells, they droop down but they look stubbier and may not be very obvious unless you clear the bees off the frame first. Be careful not to destroy naturally-made emergency cells – there won’t be more eggs available for a queen to be made.

Supersedure cells are similar to emergency queen cells. They will be found in the colony when the queen is old and unable to lay fertilised eggs, or if she is damaged. These should also be left alone.
Swarm control
If you find a swarm cell in the colony, what do you do? You can try to knock down swarm cells, but it will be hard work to keep on top of suppressing swarming and you will be working against the instinct of the bees which want to swarm in order to reproduce. They will keep building swarm cells, and they can be sneaky about it, so it is a hard and pointless fight to try to win.
A better alternative is to perform a split. Unfortunately, this requires a second set of boxes or a nuc box (a small box which holds five or six frames of bees) to split some of the brood into. The process is fairly straightforward but cannot be explained properly in this short article. Instead, I will refer you to the literature I learned from myself – Wally Shaw’s book, An Apiary Guide to Swarm Control is available through the Welsh BKA website. I highly recommend it; it’s very clear and uses easy-to-understand language. If additional colonies are not needed, they can easily be reintegrated into a single colony at the end of the season, ensuring a large population of bees – important for surviving the winter.
Pre-emptive swarm control
Last year, I split five colonies in late April. I wanted to increase the number of colonies in the apiary, so I had new boxes ready for the bees to be transferred into. Splitting a colony before swarm cells appear is sometimes called pre-emptive swarm control, in that the resulting split reduces congestion in the colony and it should reduce the urge to swarm. For this to work, you must have at least six frames of brood with a good mix of brood stages. From the six colonies I split, one of them never really got going, so I reunited it with the other colony late in the season by placing a piece of newspaper between the brood boxes to prevent the bees from taking exception to one another. This reuniting of the boxes helped to ensure the colony had a strong population and didn’t perish over the winter.
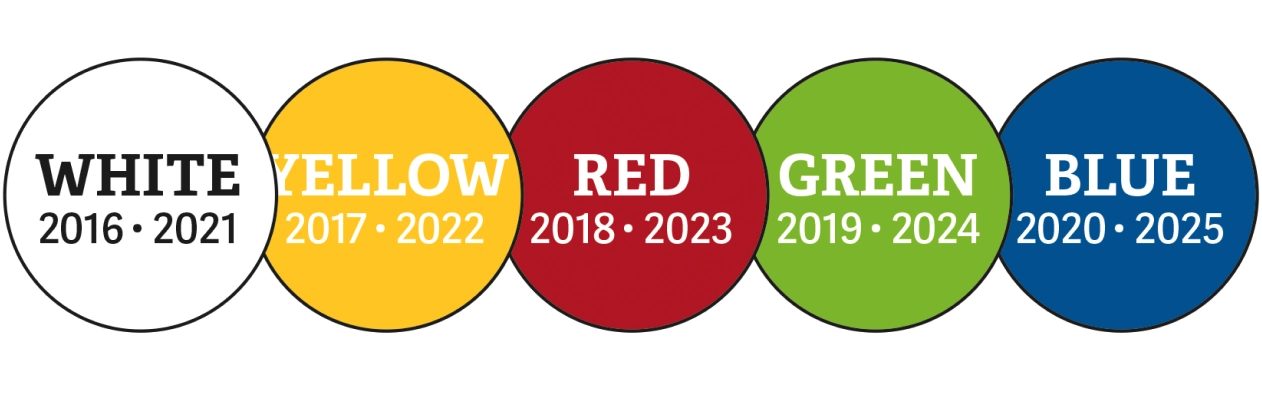
If you make new queens from your swarm control splits, you may want to consider marking them. Posca pen markers are perfect for the job as they don’t contain any harmful ingredients, and the nib is the right size for making a single mark on the queen’s thorax. If you are uncertain about queen handling, I recommend purchasing a one-handed queen catcher which will help you hold the queen securely without damaging her. I would also recommend lots of catching and marking practice with drones (see Michael Badger’s tip on page 42). The convention is to mark queen bees with a different colour each year. This is a universal standard, so if you get a marked queen from another beekeeper, you know how old she is. This year, queen bees should be marked red and this will change every year following the same pattern over a five-year period.
Money in the bank
¹Spring is the ideal time to make a few drawn frames, when the factors for wax production are present – warm weather, an abundance of young bees and a good nectar flow; bees won’t draw out wax from stored honey. Creating wax comb is costly in energy terms for the bees, so drawn-out frames are like money in the bank for future use. In Lesley Goodman’s book Form and Function in the Honey Bee, she states that it takes 1kg of honey to produce 60g of wax. Therefore, an average hive containing 1.2kg of wax has used approximately 7kg worth of honey to make it.¹
Drawn frames can be useful when splitting colonies or for comb replacement; combs should be replaced every two to three years as, over time, they accumulate traces of propolis, cocoons and faecal matter resulting in smaller-sized cells. There may also be accumulation of pathogens, or chemical residues from some varroa treatments. Whether it’s for replacing old dark combs or for expanding colonies, there is always a need for frames of drawn-out wax.
As it is so costly to produce, bees won’t make more wax than they need. Queenright colonies will draw out wax based on two criteria – the presence of a nectar flow and how much stores are present in the colony. If there is a nectar flow and a high level of filled frames, the bees will be stimulated to draw comb to accommodate the incoming nectar, but they will not draw comb if there is no nectar flow, or if there is ample space in the frames for the incoming nectar.² An abundance of young bees is also needed because those aged 10–20 days are usually responsible for wax building, as this is when their wax secretion glands are at an optimal stage of development for wax production.¹ The workers present in the hive before new bees begin emerging in early spring are practically geriatric at many months old.

How bees make wax
Wax production in honey bees is tightly regulated because it is so costly. Bees need to produce wax only when needed or they will be wasting valuable energy. Wax is made from hydrocarbons, fatty acids, and proteins called lipophorins. The worker honey bee has eight wax-producing glands between segments four and seven underneath its abdomen. As the honey bee ages, cellular changes happen in the wax-producing glands allowing the production of wax at around 7–10 days old, peaking at two weeks old before tailing off.³ Although this is a constant process, even among overwintering workers, bees aren’t constantly producing wax. The action of making wax is triggered by a flow of nectar, the current levels of stores in the colony and stimulated in the presence of the queen. Colonies with mated queens produce significantly more wax than those headed by virgin or queenless colonies. (4)

Disorder of the month
Last month, I mentioned that chilled brood can be associated with chalkbrood. Chalkbrood is a fungal disease caused by Ascosphaera apis, which affects developing honey bee brood. Chalkbrood is an opportunistic pathogen and tends to be most prevalent during the spring and early summer when colonies are growing quickly; this is when the larvae are most likely to become chilled, making them more susceptible to infection. The fungus grows best at temperatures slightly below optimal brood rearing (which is 34.5C), so this is why chalkbrood is often associated with chilled brood. Chalkbrood is fatal to infected larvae and can result in a decline in bee numbers and honey production, but it does not typically cause the demise of a whole colony.
Chalkbrood infection is easy to recognise: larvae will initially be covered by fluffy, white fungus which then dries out and shrinks to form a distinctive mummified chalk-like appearance. Like many brood disorders, there may be perforated cappings where bees have attempted to investigate what is going on underneath. Workers will uncap and remove infected chalkbrood cells, resulting in an uneven or pepperpot brood pattern. The chalkbrood mummies removed by the workers can be found on the floorboard or outside the entrance. Chalkbrood mummies are fairly distinctive but could easily be confused with mouldy pollen. However, when rubbed between two fingers the mouldy pollen will break up easily, whereas a mummified chalkbrood larva is hard to crush between the fingers.
Chalkbrood is considered highly infectious and spores can be spread through robbing and drifting of worker bees, and via contaminated equipment. Chalkbrood spores are very persistent in the environment and may last for at least 15 years. Management techniques to reduce chalkbrood infection include keeping hives clean (I regularly scrape out my brood boxes and swap out with clean boxes when necessary) and replacing brood combs at least every three years. It is important to use clean equipment and not to transfer material between colonies if infection is present, and especially not to feed honey from infected hives back to bees.
References
1 Goodman L (2003). Form and function in the honey bee. Northern Bee Books and IBRA, UK.
2 Pratt SC (2004). Collective control of the timing and type of comb construction by honey bees (Apis mellifera). Apidologie, 35: 193 – 205.
3 Cassier P and Lensky Y (1995). Ultrastructure of the wax gland complex and secretion of beeswax in the worker honey bee Apis mellifera L.. Apidologie, 26: 17-26.
4 Whiffler HR and Hepburn LA (1991). The queen in relation to wax secretion and comb building in honey bees. J Comp Physiol. A. 169.

Kirsty Stainton
Kirsty Stainton took up beekeeping in 2016 and has both practical and scientific knowledge of bees. She has worked as a senior scientist in bee health at Fera, and now keeps bees near York. With a PhD in molecular biology from Oxford University and a love of communicating science in an accessible way, her new series makes for fascinating reading.


