Techniques in the apiary: Varroa-monitoring methods
In July, 12 beekeepers attended the Advanced Beekeeping Course run by the National Diploma of Beekeeping (NDB) at Pershore College in Worcestershire. One of their exercises was to compare different methods of monitoring the number of varroa in a colony. BeeCraft was invited to observe the process and its results.
To compare varroa-monitoring techniques, the beekeepers on the course were split into four groups, each of which performed the same five monitoring methods on comparable full-size production colonies, sharing and reviewing their results at the end of the exercise. The following methodologies were used.

Natural varroa drop
Method
Clean inspection boards are placed under hives seven days before the exercise.
Remove the boards (not in windy conditions) and count the number
of mites. Magnifying equipment is useful.
Divide the number of mites by seven to give the average daily mite drop.

Drone brood uncapping
Since drones take longer to develop than workers, drone cells are the preferred target cells for the varroa mite to reproduce. Removing pupating drones can help determine the level of varroa infestation in a colony.
Method
A shallow frame is placed among the deeper frames in a brood box. Workers tend to build brood comb from the bottom of such frames, making it easy to inspect.
When drone cells are capped and mature, use an uncapping fork to uncap about 100 brood cells by piercing them with a fork and levering out the developing pupae.
Ideally, the drones should be in the later, purple-eyed stage of development, when varroa mites are also well-developed and easier to see.
Count the percentage of infected pupae, not the total number of mites (the number of infected cells divided by the total uncapped multiplied by 100).

Alcohol wash
To discover the number of mites on a sample of bees by killing mites (and bees) in an alcohol solution. Test bottles with an inner mesh container for holding and straining the bees, plus measuring scoops are available from suppliers.
Method
Prepare the alcohol mix (one part isopropanol to four parts water) and add to the test bottle.
Shake bees from selected brood combs into a large container (avoiding the queen).
Spray the bees with a fine water mist so they clump together, making them easier to control and collect.
Using a measuring cup calibrated to hold approximately 300 bees, scoop up and add the bees to the test container.
Secure the lid and gently shake the bottle to wet and kill the bees.
Shake and roll the bottle for one minute. Do so gently to avoid foaming.
Open the lid and remove the mesh basket containing the dead bees. Drain as much of the alcohol back into the bottle as you can.
Count the mites left behind in the liquid.
Divide the number of mites by three to give a percentage of mite infestation in the hive (assuming there were originally 300 bees).
For a more accurate result, count the dead bees to calculate a precise percentage.

CO2 method
A relatively new technique and good for those who don’t want to kill bees – although the equipment is more expensive. A sample of bees is anaesthetised with CO2 while any varroa, also anaesthetised, are shaken from them. The bees are returned to the hive where they quickly revive. The test equipment is available as a kit from suppliers.
Method
Shake bees from brood frames into a large container, as in the alcohol wash method.
Scoop approximately 200 bees into the test pot. There is a line on the pot to show when 200 bees have been collected.
Place the plastic lids on both ends of the pot and turn it so that the bees are at the bottom, below the mesh divider.
Insert the nozzle of the CO2 dispenser and inject the gas for 4–6 seconds.
After 10–20 seconds, shake the anaesthetised bees gently for 10 seconds, The dislodged mites will fall through the mesh.
Open the lid to allow fresh air to the bees, tipping them back into the hive, where they quite quickly revive.
Remove the lid at the bottom of the pot and count the mites that have fallen through the mesh onto it. Divide the number by two to give the percentage.

Icing sugar method
Another method that is not fatal to the bees – and even gives them a sugary treat to compensate for the disturbance. Bees are shaken in icing sugar, causing varroa to lose their grip and fall off to be counted. The sugar-coated bees are returned to the hive, dazed but unharmed.
Method
Use any container that has a mesh or perforated lid that will allow icing sugar but not bees to pass through. Beekeeping suppliers sell suitable containers.
Collect 300 bees (avoiding the queen).
Add about 7g of fine, dry, icing sugar.
Roll the jar gently so that the sugar coats the bees.
Let the jar stand for one minute.
Invert the jar and shake over a white surface until all the icing sugar and no more mites fall through. This is best done indoors if breezy.
Count the mites and work out the percentage.
Return the bees to the hive.
Results and conclusions
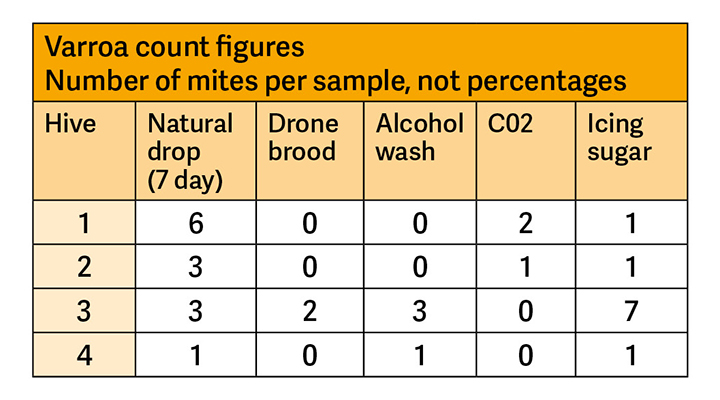
At the end of the exercise, the NDB students returned to the classroom to compare results and discuss the methods. Relatively few varroa were found and this was considered a credit to the beekeeper at Pershore, Rhona Toft, who has long selected for varroa-coping abilities in her bees and has not treated them since 2016.
It was felt that on this occasion the alcohol wash and icing sugar methods were the most effective at separating varroa from a sample of bees. Some people were uneasy about the alcohol method because it killed the bees, although it was considered that the loss of 300 bees would not adversely affect a healthy colony. However, killing the bees does allow them to be carefully counted, giving a more accurate indication of the sample size than with other methods. Some of the group used the camera and magnifying function on their phone to help with the count. The alcohol wash method is slightly more expensive because it uses isopropanol instead of icing sugar. Both methods were considered to be more effective and better value than the CO2 method, which uses relatively costly equipment and materials. The advantage of all three methods is their efficiency, allowing varroa levels to be monitored quickly and at short notice.
Bob Smith NDB, one of the tutors on the course and formerly a research chemist, stressed the importance of high-quality sampling. With one exception, each of the tests used samples of bees gathered from a small part of the colony – typically a single brood frame.
The natural drop method, on the other hand, counts mites falling from bees on all of the frames in a hive and over an extended period, therefore probably providing a more representative sample. Indeed, this method indicated the presence of varroa in every colony when in some cases other techniques produced no evidence at all.
It was suggested that the pattern of mites falling from a colony can change depending on the activities of the bees (during a flow they might spend less time preening, for example), so using the natural drop method frequently and for a minimum of seven successive days will give a more balanced result. Course tutor Lynfa Davies pointed out that various predators can remove fallen varroa, so it is worth counting the fallen mites and cleaning the board several times, adding the counts to get a total for the seven days. Overall, the natural drop method was considered one of the more effective techniques.
The drone brood uncapping technique was considered effective but required the most skill, both in identifying brood of the right age and in levering it out of the comb. One advantage is that this technique can be combined with a biotechnical varroa control method, cutting the whole the slab of drone comb from below a brood frame to remove the varroa it contains from the colony.
Interpreting the results
There was considerable discussion about how the varroa counts indicated infestation levels or the need to treat a colony. Commercially available equipment usually comes with instructions indicating various thresholds, although they are somewhat variable.
The simplest results to interpret were from the drone brood uncapping and natural mite drop techniques since both methods are recommended by the National Bee Unit (NBU) and details of the methods and how to analyse and act on results are given on its BeeBase website (www.nationalbeeunit.com).
The overall conclusion was that varroa monitoring is not very rigorous and it that it probably doesn’t matter which method you use. The key is to monitor colonies consistently compare like-for-like results to give a sense of varroa infestation as it changes throughout the season.

The NDB Advanced Beekeeping Course
An annual five-day residential course held at Pershore College in Worcestershire, it is designed for progressive beekeepers looking to improve their skills and knowledge and to prepare future candidates for the NDB examination. Course tutors are Lynfa Davies and Marin Anastasov plus guest speakers.
Students undertake classroom activities, lab sessions, practical apiary work and relevant workshops with a three-year rolling programme to cover a wide range of beekeeping topics. Highlights this year included a lab session on botany to help students understand how plants work. This was enhanced by a session on plant families and plants relevant to bees. There was a practical session about identifying bumblebees with expert entomologist Liam Olds, and a lab session with diseased combs to give the students exposure to recognising the foulbroods in combs provided by the NBU.

Richard Rickitt, Co-editor
Richard Rickitt is an award-winning author and co-editor of BeeCraft, the UK’s best-selling beekeeping magazine, read by beekeepers and entomologists around the world. He has been keeping bees for more than twenty years and keep hives for a number of commercial and private clients. Richard is beekeeper at Westonbirt, the National Arboretum, and he teaches beekeeping courses in the UK and abroad.


